 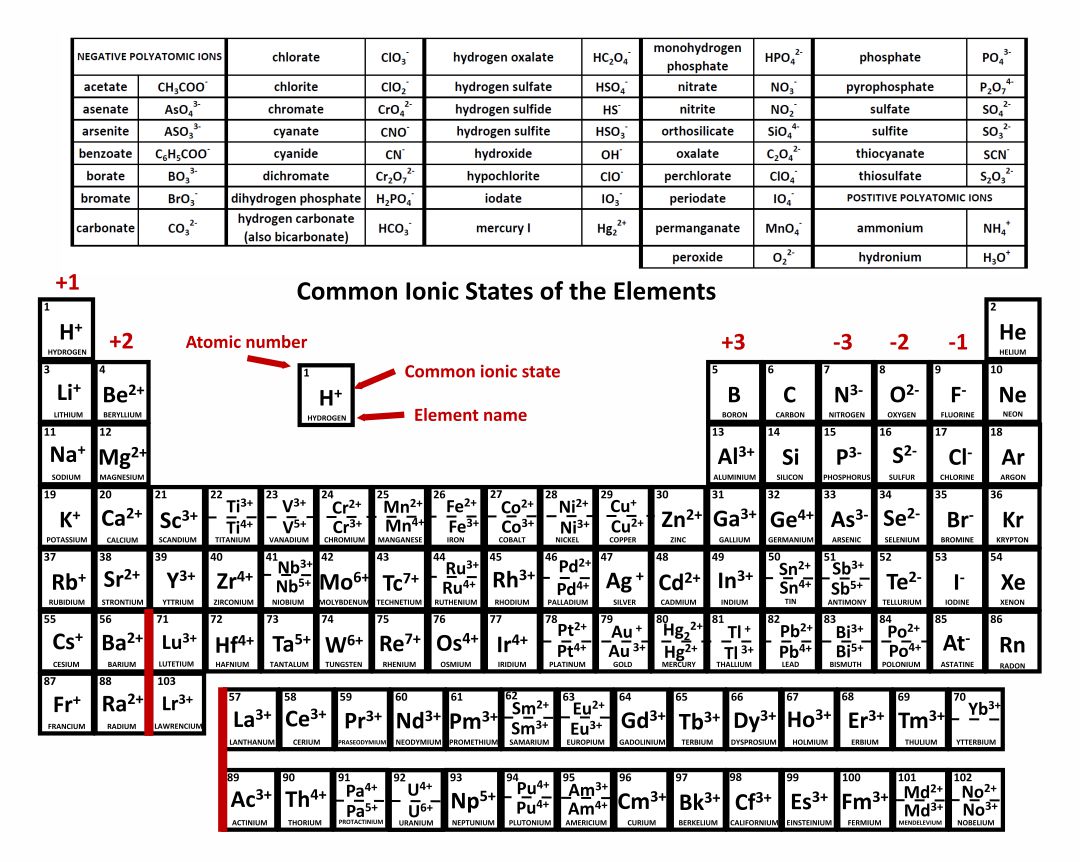
This is to show that the subscript applies to the entire polyatomic ion. If more than one of a particular polyatomic ion is needed to balance the charge, the entire formula for the polyatomic ion must be enclosed in parentheses, and the numerical subscript is placed outside the parentheses. Some ions consist of groups of atoms bonded together and have an overall electric charge. The periodic table, its story and its significance. A neutral molecule that has an area of positive charge and an area of negative charge is called a zwitterion. An ion with a +2 charge is a cation that is also called a dication. The most-requested printable periodic table lists element charges, to predict compounds and chemical reactions. The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. The naming of ionic compounds that contain polyatomic ions follows the same rules as the naming for other ionic compounds: simply combine the name of the cation and the name of the anion. An ion with a -2 charge is an anion that is also called a dianion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
